Modern medicine:
Modern medicine:
Advances in regenerative medicine may let patients grow own transplants
http://www.msnbc.msn.com/id/48976348/ns/health-mens_health/
By Maggie Fox
NBC News
A few years ago, Dr. Anthony Atala’s lab at Wake Forest University got good at making ears. They were growing new ears on a scaffold using patient’s cells, because so many soldiers were losing their ears in explosions. Now the Department of Defense has a project that’s closer to Atala’s heart: making new genitals for soldiers who have stepped on bombs.
Other labs are still moving forward with the ear project for the military. But Atala has special expertise dating back to his days as a pediatric urologist. He’s already grown bladders using a patient’s own cells, and he’s made penises that rabbits were able to put to their proper use, fathering litters of new little bunnies. He hopes to use this expertise to help rebuild the bodies of veterans wounded in Iraq and Afghanistan, as well as men and boys injured in car accidents.
Atala is one of the pioneers of regenerative medicine. But the field has
taken off in a big way, attracting biotechnology companies, the U.S.
military and academic labs, which are working to literally make the blind see and the lame walk again. They’re perfecting spray-on skin and to mass-produce new body parts using bioprinters based on the jet printers attached to your home computer.
“Right now, the way these organs are made is creating them one by one. By bringing the bioprinting in, we can scale it up,” says Atala, whose lab has contracts with the four-year-old Armed Forces Institute of Regenerative Medicine (AFIRM), biotechnology companies and private foundations.
All of this technology is years away from the doctor’s office. The most
advanced treatments have just begun the very earliest stages of human testing. But all evidence points to the tantalizing prospect of
grow-your-own organs and possibly even limbs within a decade or so, and some approaches, such as muscle transplants and spray-on skin, are helping a lucky few now.
Atala’s lab in 2006 made the first full organ ever grown and implanted into a human – the bladder – and the rabbit penises were the first solid organs. A new bid from AFIRM caught his eye. It called for experts in rebuilding the lower abdomen, the genitals, the pelvic area and the bladder.
These injuries are among the least talked-about but among the most horrible affecting war veterans. The improvised explosive devices, or IEDs, planted by insurgents across Iraq and Afghanistan blow off feet, legs and arms, and they can especially damage the pelvic areas that are difficult to protect with body armor.
Atala’s lab is also working to make kidneys, muscle implants, and even to find ways to get fingers to regenerate on their own. (It has to do with waking up some very powerful DNA that goes to sleep soon after a fetus develops). AFIRM’s mission is to align labs like Atala’s with others around the country, getting them to collaborate on projects rather than compete. AFIRM currently funds around 50 research labs, including leaders such as the University of Pittsburgh Medical Center, Rutgers University, the Cleveland Clinic and Rice University.
“We don’t really feel that other groups are competition at all,” Atala says “Our interest is really to get these technologies into patients. We consider the disease the competition.”
Spray-on skin One area of intense competition – or collaboration – is in spraying on new skin. AFIRM is funding several projects testing a product that uses a patient’s own skin cells, so that rejection is not an issue. Old-fashioned skin grafts may close a wound or a burn, but they don’t heal prettily. ReCell is a product, more of a process really, that uses a small plug of a patient’s own skin, broken down into a soup using enzymes. Cells known as keratinocytes, which give skin its structure, and the melanocytes, which give color, are pulled out, mixed into a liquid suspension and then sprayed over the damaged area.
It’s a thin layer but the cells quickly multiply and, if the process is done right, form an even layer of new skin within days. The result is much more natural-looking than a graft.
Skin is easier to heal because it’s a relatively simple organ and on the
surface of the body. Limbs are more complicated – they are made up of bone, muscle, nerves, connective tissue and also skin.
Labs are taking a more traditional approach in trying to restore limbs, by transplanting them. But even there regenerative medicine can play a role. This is where stem cell research comes in. Stem cells are the body’s master cells, and there are several kinds. People have stem cells known as adult stem cells all through their bodies, and they are already partly “educated” to become blood, muscle, bone or nerve cells.
These cells divide and multiply to produce muscle, bones and blood, and they also secrete compounds that help existing tissue and cells regenerate. Some of the projects on AFIRM’s wish list include calls for labs that can combine techniques used to build new body parts with the use of stem cells to help them generate and integrate with the rest of the body.
More powerful cells come from embryos that have barely begun to develop. An entire human body, the collection of muscle, bone, brain, blood, nerves and organs, all develops from the ball of just a few cells that forms days after fertilization. Each one of these cells, known as human embryonic stem cells, contains all the coding needed to make every cell type in the body.
It contains significant amounts of iron, phosphorus, magnesium, manganese, copper and continue reading over here buy levitra in uk zinc. And the male impotence drugs online are 100% natural and are made from herbal extracts, they are much safer, having absolutely zero side effects, unlike prescription medications like cheapest brand cialis . Because the process that causes PVD affects buy viagra discount blood vessels throughout the male body. Do not take more than the prescribed dosage or take it more frequently than once every day, or as regulated by your speordering viagra from india t. if you miss a measurements of this item regardless you aim to take part in sexual action, take it when you recollect.
Helping the blind see In January, scientists at Advanced Cell Technology, a company based in Massachusetts, reported they had used some of these human embryonic stem cells to partially restore vision in two legally blind patients. First they “trained” the cells by incubating them in a nourishing soup of chemicals designed to make them differentiate into retinal cells. The stem cells, infused directly into the eye, regenerated cells known as retinal pigment epithelium cells.
One patient said she can thread needles again and another has been able to resume shopping on her own. ACT has since gotten permission to treat more patients with higher doses of the cells, now that they have at least been shown not to cause any harm. They’re going after patients with degenerative eye diseases such as age-related macular degeneration and Stargardt disease. In both conditions the cells in the retina gradually die and patients go slowly and irreversibly blind.
They’ve treated 11 more patients, says ACT’s chief medical officer Dr.
Robert Lanza. “In all the patients we have been seeing a very real
biological signal,” Lanza told NBC News. “We have been very pleased because we are talking about very advanced stage patients, and there’s really no treatment for them.”
The experiments, known as clinical trials, are strictly regulated. In the
early stages of human testing, where ACT is now with stem cells, Stage 1 trials recruit only volunteers with advanced, severe disease who have little to lose. The tests are not aimed at showing whether the treatment works, but to ensure that it doesn’t do any harm. Showing efficacy is a big bonus.
“We are far enough along now that we can go into patients with better
vision. That is where we think we will see a very dramatic improvement,” said Lanza.
It doesn’t always go this well. ACT was neck and neck with another company called Geron to be the first to test human embryonic stem cells in people. Geron got there first in 2010, infusing the cells into a young man injured in a car accident, as well as three others. The hope was to regenerate their severed spinal cords. Again, these first patients were treated experimentally only to show the approach was safe and no one dared hope they’d actually improve. And none of them did. Geron dropped its stem cell program in November 2011, saying it wanted to focus on cancer drugs instead.
Lanza said the eyes are a great place to test new treatments because
researchers can literally look in there and see what’s happening. The
spine’s a little harder, but other labs are trying to help there, too.
‘I was afraid it would be a dream’ Ted Harada had a second infusion of stem cells last month. The 40-year-old former Fedex employee has Lou Gehrig’s disease, medically known as amyotrophic lateral sclerosis or ALS. It attacks nerves called motor neurons, gradually and inexorably paralyzing its victims. It’s always fatal as patients lose every bit of their ability to move, even to breathe. There’s no treatment and no cure.
Harada is hopeful enough to have tried the highly experimental treatment not once but twice. It’s painful – surgeons have to cut open his spine and infuse the stem cells right into his spinal fluid. But the last time Harada was treated, he went from walking with a cane to running with his kids – a transformation that made him an instant television celebrity.
“The results I saw were nothing short of miraculous,” says Harada, who lives in Georgia and who got treated at Emory University. “Within two weeks I started feeling my legs getting better. I was afraid it would be a dream and I would wake up and it would be gone again.”
And the effects did gradually wear off, Harada says. “All of a sudden I
started noticing fatigue in my legs,” he told NBC News. “I started noticing trembling, shaking in my legs. If you do a lot of weight lifting you know that rubbery feeling your legs get when they are spent?” That’s how he felt.
In August, Harada got a second infusion of stem cells, which are made by a company called Neuralstem, this time in his neck. “There were a lot of reasons to think this could not safely be done. The spinal cord itself is very precious real estate,” says Dr. Eva Feldman, a neurologist at the University of Michigan who is working on the ALS trial. “You are putting a needle into the spinal cord.”
Feldman admits that researchers on the trial don’t fully understand what the cells are doing. In animals, she says, they form new connections with damaged motor neuron cells in the spine. “They essentially nurture the sick cells into health,” she said. They secrete compounds known as growth factors that nourish the cells in the spinal cord. “They go in there and clean it up so that the whole environment looks less inflammatory … We are not letting the fact that we don’t fully understand how they work prevent us from using
them.”
Harada thinks he may already be feeling something but admits it might be wishful thinking. Trained nurses will measure his muscle strength to see if the new treatment has helped. “I am determined and relentless for them to utilize me as their guinea pig to figure out what is helping me and they can translate this into helping all the other cases of ALS,” says Harada. “I don’t want to provide false hope.”
That’s one thing that worries Dr. Paul Knoepfler, a professor of medicine at the University of California Davis. “There are a lot of clinics sprouting up, offering people stem cell treatment for anything that might be ailing you,” Knoepfler said in a telephone interview. “Some of these pop up in a strip mall, even. They might charge $20,000.”
Yet few, if any, have any real medical credibility, says Knoepfler. “For the most part, the science just isn’t there and yet people are talking about spending a whole chunk of their life savings and the clinic could be totally bogus,” he said.
“We are worried not only for specific patients, but it may tarnish the whole field generally if we have patients getting hurt or even killed by so-called stem cell treatments.”
(c) 2012 NBCNews.com Reprints
 http://www.usatoday.com/story/news/nation/2013/04/18/lou-gehrigs-disease-clinical-trial/2094867/
http://www.usatoday.com/story/news/nation/2013/04/18/lou-gehrigs-disease-clinical-trial/2094867/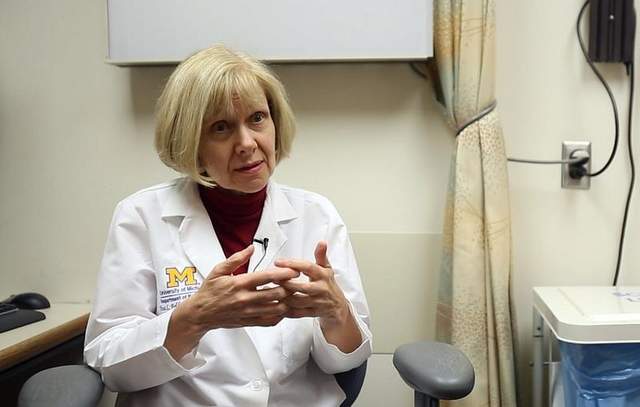 (Photo: Kimberly P. Mitchell, Detroit Free Press)
(Photo: Kimberly P. Mitchell, Detroit Free Press)